Reading Theme:
Oxide Definition, Common Uses & Chemical Properties
Meaning 1:oxygen compound (oxide)
oxide
/ˈɒksaɪd/
n.
A compound formed when oxygen combines with another element or group.
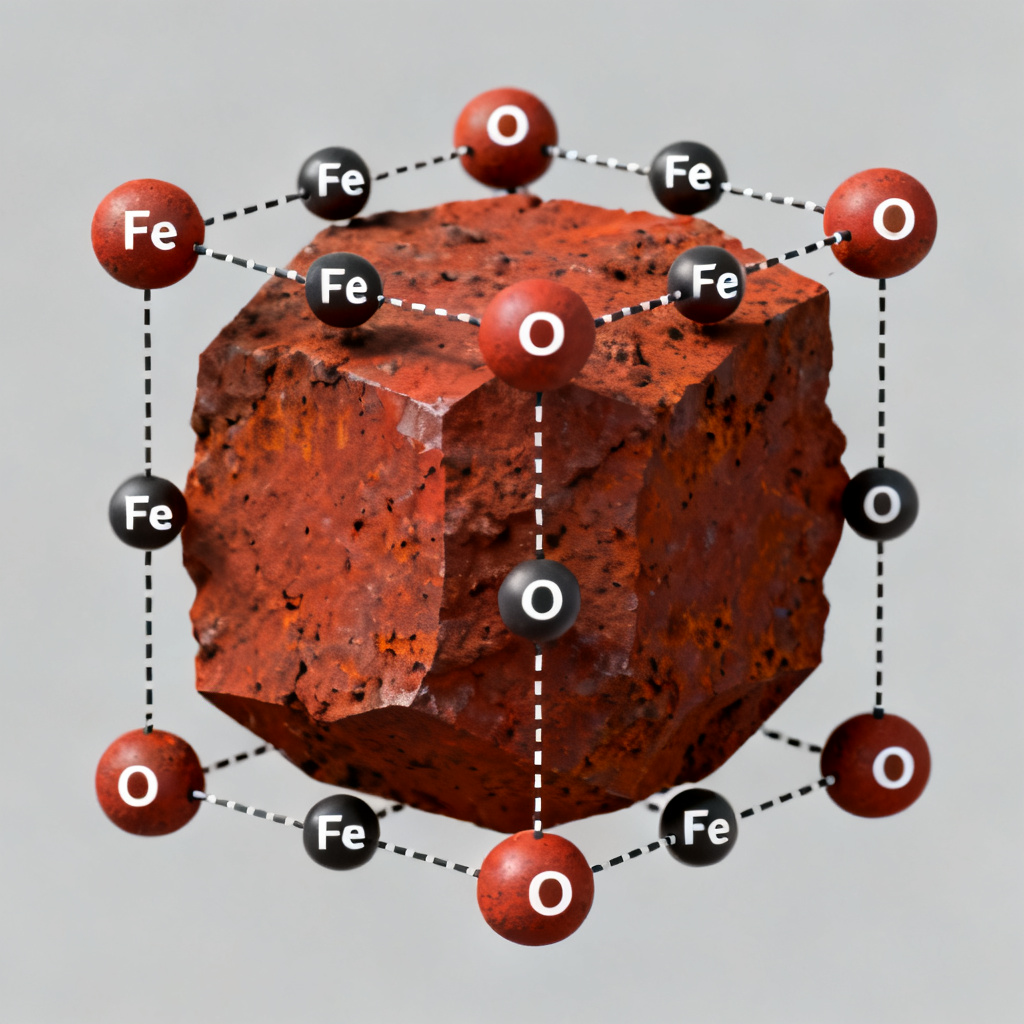
oxygen compound
➕
/ˈɒksɪdʒən ˈkɒmpaʊnd/
n.
A chemical substance where oxygen is chemically bonded to another element or group, often resulting from reactions like corrosion.
📘 Details & Usage
📖 Root Explanation
From Greek 'oxys' (sharp, acid) + '-ide' (suffix for compounds).
💡 Mnemonic
Think: Oxygen + ide suffix = oxide, a compound containing oxygen.
📖 Example
When iron is left outside in the rain, it slowly turns into oxide, which we commonly call rust.
When iron is exposed to rain, it gradually transforms into oxide, commonly known as rust.
🔗 Collocations
iron oxide – a compound of iron and oxygen, often seen as rust
aluminum oxide – a compound of aluminum and oxygen, used in industrial applications
nitrogen oxide – a compound of nitrogen and oxygen, frequently an air pollutant
🔄 Synonyms
rust (n.) – specifically iron oxide formed through corrosion of iron
compound (n.) – a substance formed by the chemical combination of two or more elements
corrosion product (n.) – a substance resulting from the deterioration of materials, often oxides
🚫 Antonyms
element (n.) – a pure substance that cannot be broken down into simpler substances by chemical means
pure metal (n.) – a metallic substance in its uncombined state, without oxide layers
🌱 Derivatives
📖 Cultural Story
The term 'oxide' derives from Greek 'oxys' meaning 'sharp' or 'acid', as oxygen was initially obtained from acids. It entered English via French in the late 18th century.
Wordbook
Your data is saved in this browser